Quantitative differences in samples of 300,000 IU/ml assayed diluted... | Download Scientific Diagram
Servizio “Sanità, lavoro e politiche sociali” Codice sito: 4.10/2022/25 Al Ministero della salute Gabinetto gab@postacert.s

HCV and VSV-G neutralizing activity and HCV RNA levels (IU/ml) were... | Download Scientific Diagram

Real time amplification of HCV RNA by real time RT-PCR assay. Shown... | Download Scientific Diagram
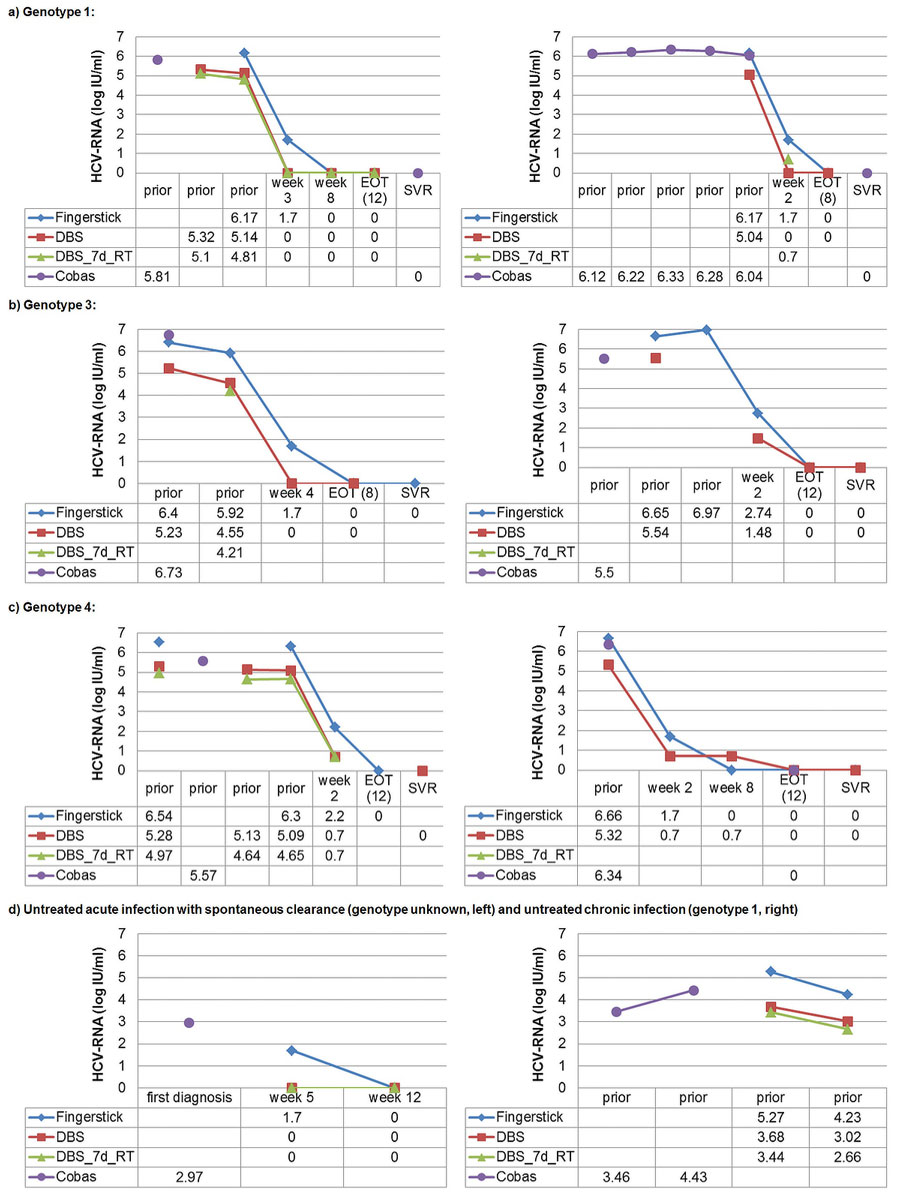
HCV RNA quantification in capillary dried blood spots with the Xpert® HCV Viral Load test for diagnosing chronic HCV infection, monitoring treatment and detecting reinfection