Crystal structures of (a) body-centered-cubic (bcc), (b) and (c) A15... | Download Scientific Diagram

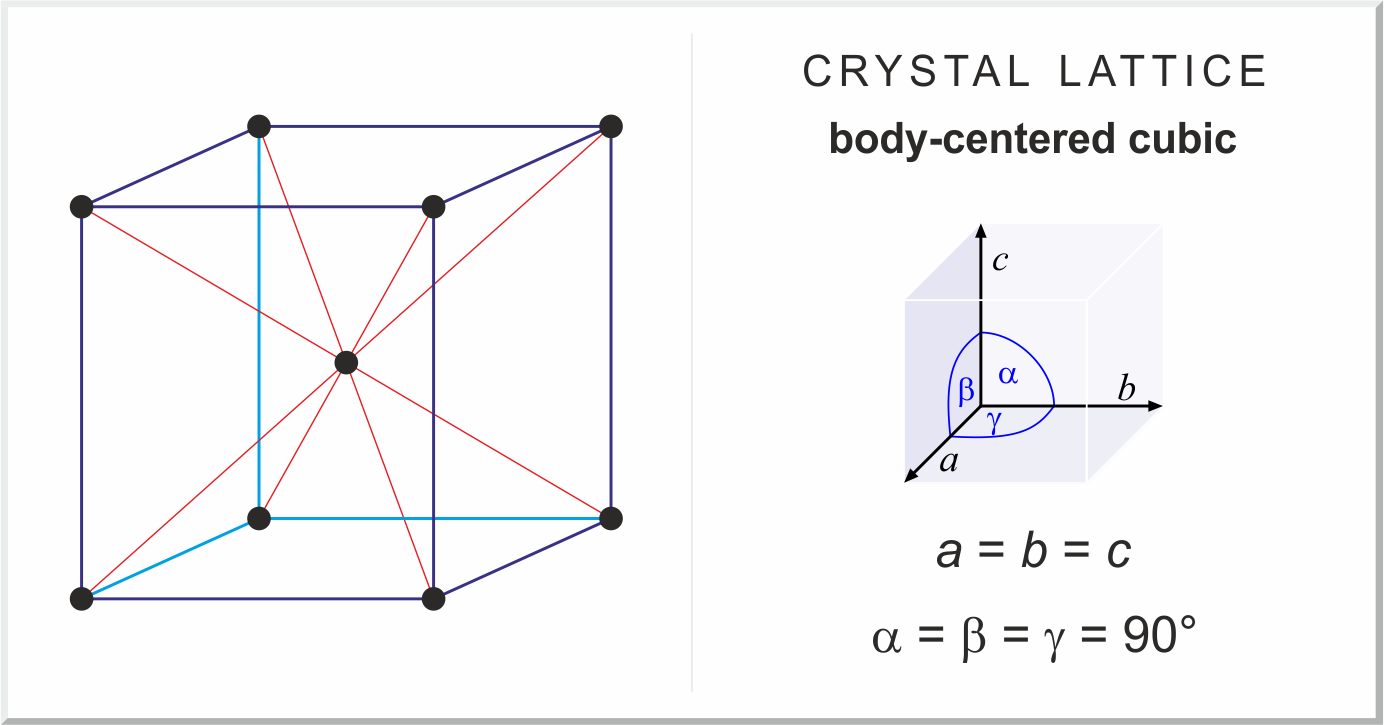
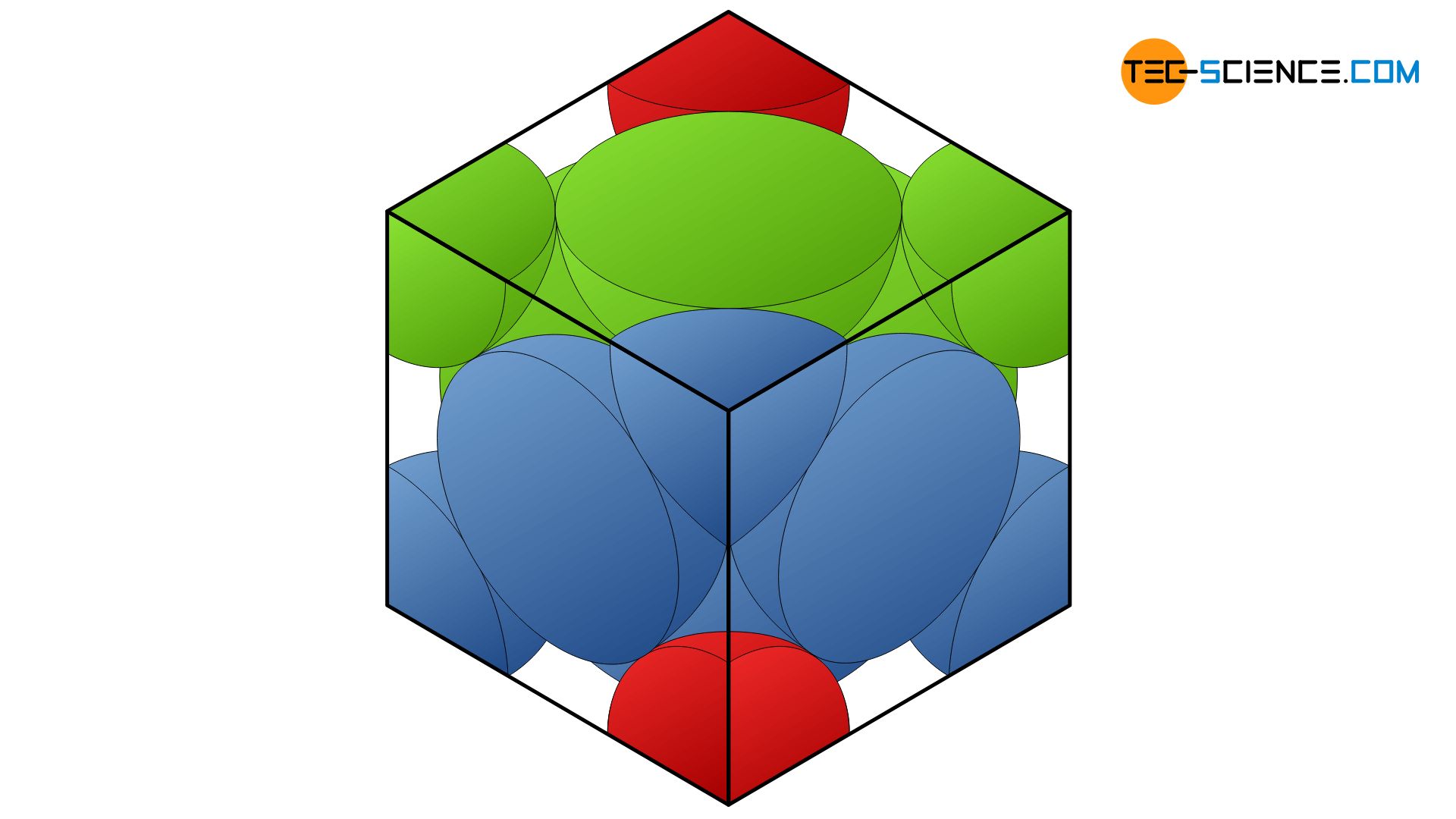
Consider a body centered cubic (BCC) arrangement, let d_e, d_fd, d_bd be the distances between successive atoms located along the edge, the face diagonal, the body diagonal respectively in a unit cell.
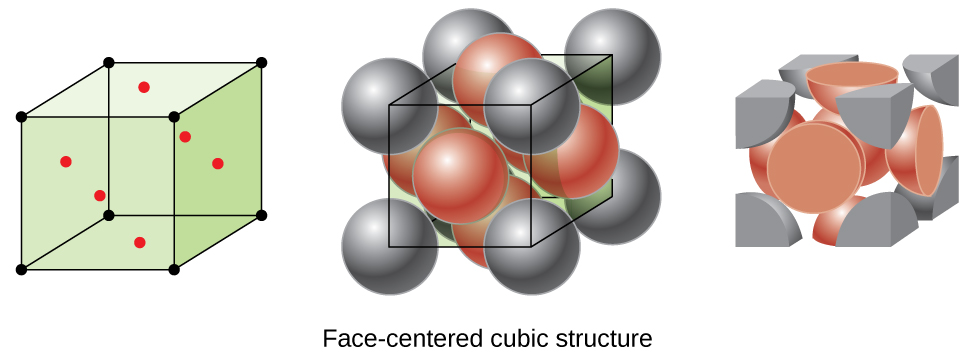
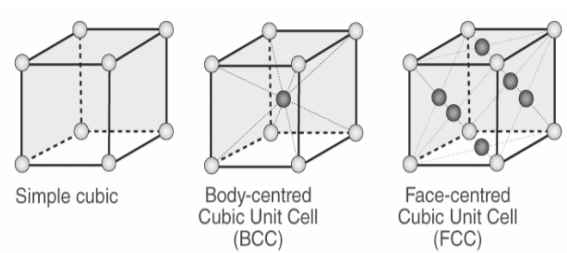
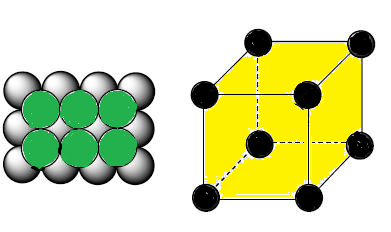
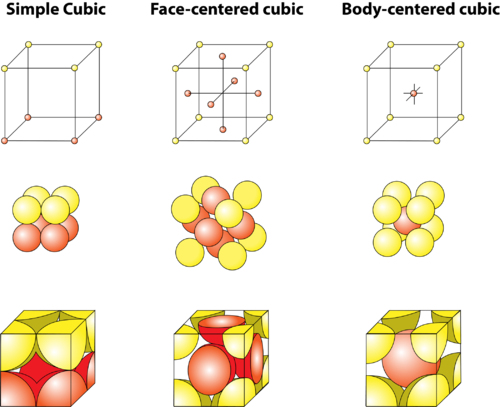
Types of Unit Cells: Body-Centered Cubic and Face-Centered Cubic (M11Q5) – UW-Madison Chemistry 103/104 Resource Book
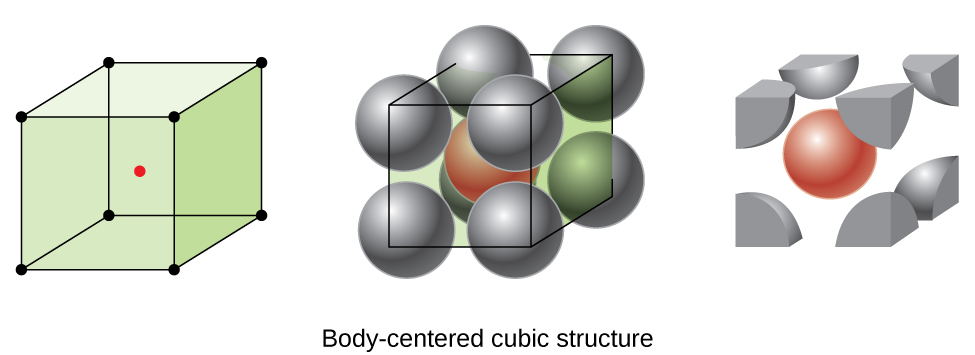
Types of Unit Cells: Body-Centered Cubic and Face-Centered Cubic (M11Q5) – UW-Madison Chemistry 103/104 Resource Book
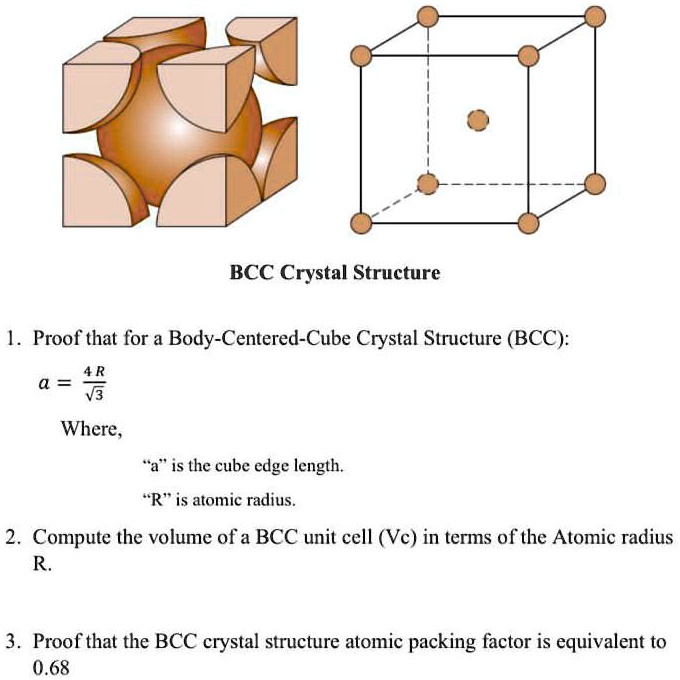
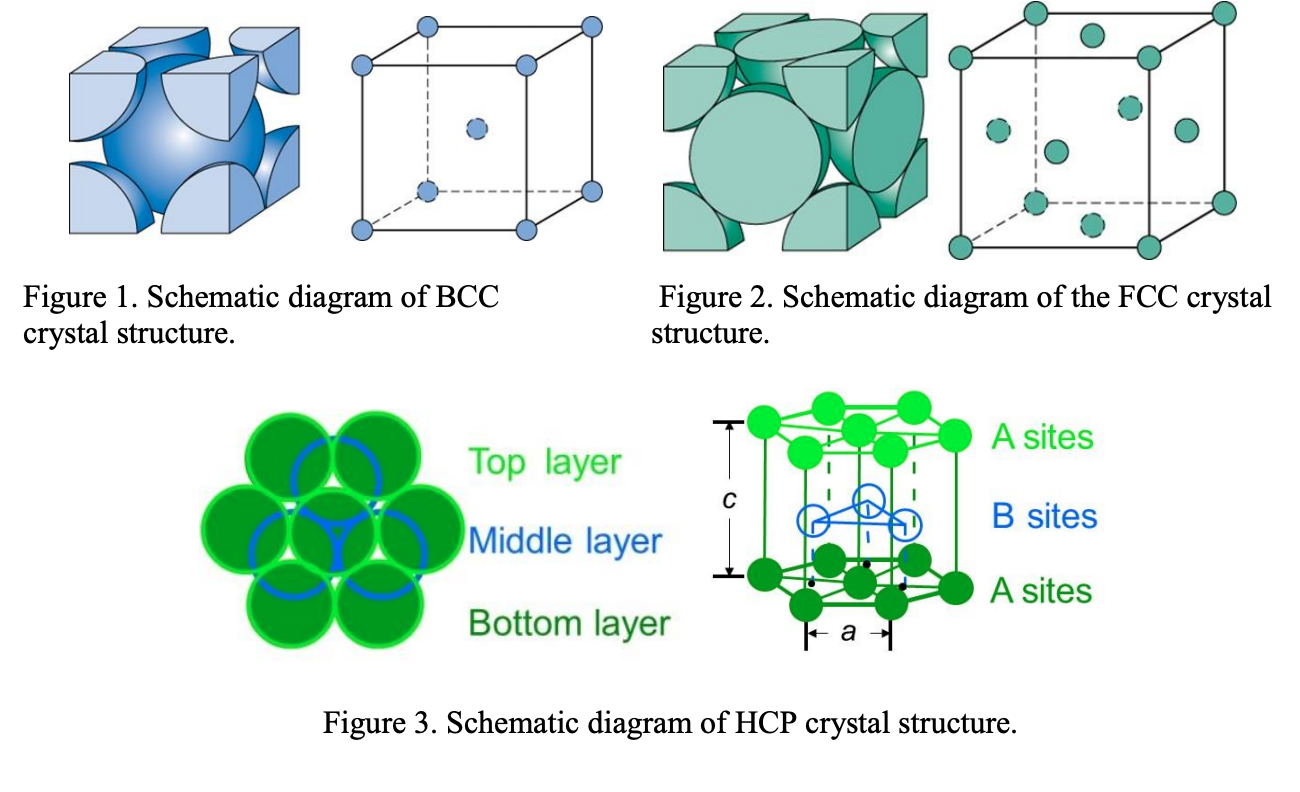
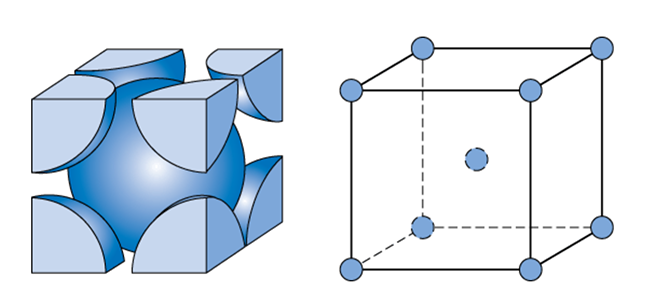
SOLVED: Proof that for a Body-Centered-Cube Crystal Structure (BCC), a = √3R Where "a" is the cube edge length and "R" is the atomic radius. Compute the volume of a BCC unit